The CHARM Trial: CHemotherapy for Ablation and Resolution of Mucinous Pancreatic Cysts
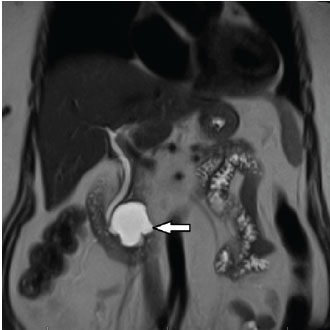
76-year-old female with a 4.2cm mucinous (premalignant type) pancreatic cyst (white arrow) prior to EUS-guided chemoablation in the Penn State CHARM trial.
Pancreatic cysts are incidentally detected in 5-20 percent of patients over the age of sixty-five who undergo CT or MRI imaging, and the majority of these cysts are comprised of premalignant type mucinous cysts. While their detection may lead to early treatment or prevention of pancreatic cancer, effective and safe management poses a challenge. “Current guidelines,” explains Matthew T. Moyer, MD, of Penn State Hershey Gastroenterology and Hepatology, “call for ongoing radiographic surveillance or surgical resection of these premalignant lesions; however, both options have significant limitations, risks, and costs.” Pancreatic surgical resection is associated with a 2 percent risk of mortality and an estimated 30 percent risk of serious complications.
Exploring less invasive, yet effective options for treating premalignant pancreatic cysts, Penn State Hershey has launched the CHemotherapy for Ablation and Resolution of Mucinous Pancreatic Cysts (CHARM) trial,1 building on previously described endoscopic ultrasound (EUS)-guided ethanol lavage for pancreatic cysts (the EPIC trial),2 and ethanol lavage followed by infusion of paclitaxel.3
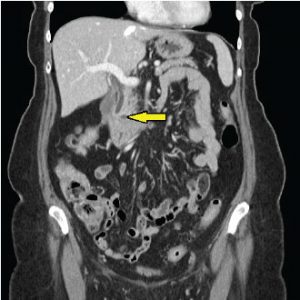
A 5mm residual defect at one year (yellow arrow); the patient will return per protocol in one year for a surveillance MR-MRCP to assure resolution.
“EUS-guided chemoablation using ethanol was a major therapeutic advance. A drawback, however, is 3-4 percent of patients developed pancreatitis (with the use of ethanol) and there was limited efficacy,” explains Moyer.
This trial aims to evaluate the efficacy and safety of alcohol-free chemoablation and proposed additional efficacy with use of a pancreatic neoplasia specific chemotherapy cocktail. Patients are randomized to EUS-guided lavage with either normal saline or ethanol followed by infusion of a custom designed ablation cocktail of paclitaxel/ gemcitabine in both groups. Moyer notes, “Gemcitabine has been the backbone of pancreatic chemotherapy for thirty years, and is one of the few chemotherapeutic agents with a proven survival benefit in pancreatic cancer. Additionally, we have shown it to be compatible and stable when used as an infusion admixture with paclitaxel. “
Post-treatment assessment is performed at seventy-two hours, with imaging at three, six and twelve months. Moyer and colleagues presented the preliminary data, based on the first nine patients to receive treatment.1 “At the three-month follow-up, identical cyst size reductions were seen with saline versus ethanol lavage, suggesting that ethanol isn’t necessary for lesion ablation, if lavage is followed by paclitaxel/ gemcitabine. One patient in the ethanol group developed self-limited pancreatitis,” says Moyer.
The new protocol promises to be a step toward improving outcomes and decreasing risks in treating premalignant pancreatic cysts. To date, twenty-three patients have been randomized and treated. Firm conclusions will require the full patient sample (seventy-eight) followed for one year.
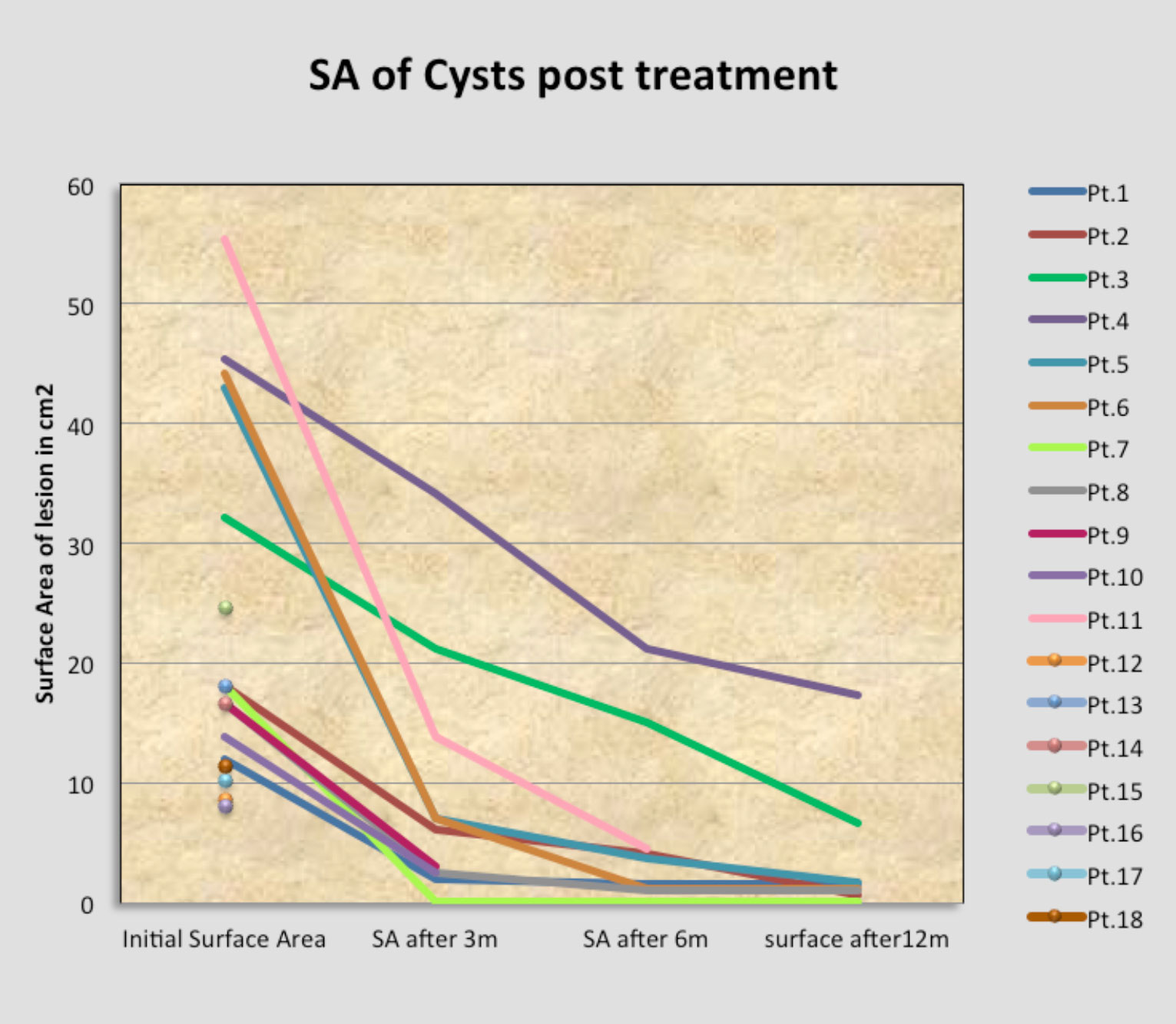
Pancreatic cyst surface area 3, 6 and 12 months post treatment to date.

Matthew T. Moyer, MD, MS
Professor, Department of Medicine, Division of Gastroenterology and Hepatology, Penn State Cancer Institute
Phone: 717-531-4950
Email: mmoyer@pennstatehealth.psu.edu
Fellowship: Gastroenterology, Penn State Health Milton S. Hershey Medical Center, Hershey, Pa.
Residency: Internal Medicine, Penn State Health Milton S. Hershey Medical Center, Hershey, Pa.
Medical School: Penn State College of Medicine, Hershey, Pa.
Connect with Matthew T. Moyer, MD, MS, on Doximity
References
1. Moyer MT, Sharzehi S, Dye CE, et al. The CHARM Trial (CHemotherapy for Ablation and Resolution of Mucinous Pancreatic Cysts): the initial pilot study for this prospective, randomized, double-blind, single-center trial [poster]. Presented at: Annual Digestive Disease Week, Chicago, IL, May 3-6, 2014.
2. DeWitt J, McGreevy K, Schmidt CM, Brugge WR. EUS-guided ethanol versus saline solution lavage for pancreatic cysts: a randomized, double-blind study. Gastrointest Endosc. 2009 Oct;70(4):710- 23.
3. Oh HC, Seo DW, Song TJ, et al. Endoscopic ultrasonography-guided ethanol lavage with paclitaxel injection treats patients with pancreatic cysts. Gastroenterology. 2011 Jan;140(1):172-9.

