Problem: How to Improve Bone Graft Integration and Strength
Solution: an elegantly “simple” biomimetic film coating.
Critical-size bone defects are challenging to bridge and successfully treat. A research team at Penn State Bone and Joint Institute, led by Henry J. Donahue, PhD, postdoctoral fellow Alayna Loiselle, PhD, and Akhlesh Lakhtakia, PhD, the Charles Godfrey Binder Professor of Engineering Science and Mechanics, Pennsylvania State University, is exploring new biotechnologies to enhance bone integration in such patients. Donahue explains, “While traditionally used allographic bone implants can be effective for bridging critical-size defects, patients encounter postoperative complications, such as ongoing pain, increased risk of bone fracture and infection at the implant site, and need for additional surgeries. Allograft bone often is not well integrated into host tissue and lacks the strength of native bone.”
Part of the problem seems to arise from the processing of cadaveric bone, which removes all host cells and disease particles prior to implantation. While helping to ensure safety, the process, however, destroys the naturally rough, nanotopographic features of the bone’s outer surface, rendering it less osteoconductive, thus hindering cell migration to the repair site and discouraging osteoblast differentiation.
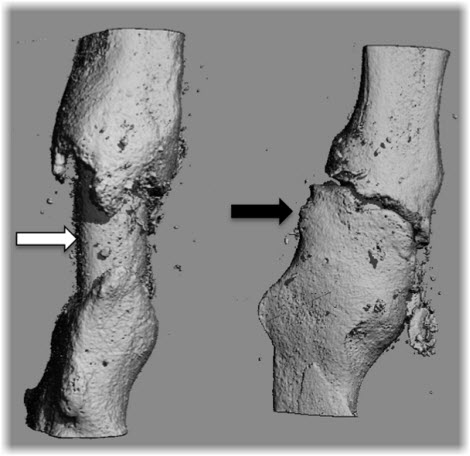
Uncoated allograft (white arrow) showing poor osteointegration and healing. Right: Fracture callus completely covering allograft coated with hydroxyapatite biomimetic film (black arrow), demonstrating dramatically enhanced bone healing. [Click to enlarge photo for detail]
In collaboration with Donahue and Lakhtakia, Loiselle has developed a hydroxyapatite-based film that exhibits an optimal nanotopography and a surface chemistry that is similar to native bone. In a recent proof-of-concept series of in vivo investigations using a mouse bone defect model1, Loiselle demonstrated that repair with allograft material coated with the hydroxyapatite biomimetic film dramatically increased callus formation and bone strength, compared to repair using uncoated allograft (illustrated in figure above). Donahue notes that, “Unlike other, newer graft materials, allograft bone coated with the biomimetic film doesn’t contain any cells or growth factors. This means it’s inexpensive to produce, has an unlimited shelf-life, isn’t immunogenic, and doesn’t require any specialized storage facilities, such as refrigeration, so that it can be used directly at the point-of-service. This is a particular advantage when surgery needs to be done in remote situations, such as on the battlefield.”
Although much work remains to be done before the biomimetic film becomes available clinically, its use in other orthopaedic applications is already being considered. Donahue speculates “The nanotopographically optimized film might be applied to other orthopaedic and dental implants that are currently in wide use, potentially helping to increase bone ingrowth, lengthen the lifespan of the implant, and reduce the need for replacement surgery.”
If the biomimetic film succeeds in clinical trials, it very well may be an elegantly “simple” solution to a costly problem that adversely affects thousands of implant patients every year.
Reference
1 Loiselle AE, Wei L, Paul E., et al. Specific biomimetic hydroxyapatite nanotopographies enhance osteoblastic differentiation and bone graft osteointegration. Tissue Engineering In Review.

Henry Donahue
Professor Emeritus
Penn State College of Medicine
Penn State Orthopaedics and Rehabilitation
Phone: 717-531-4819
Graduate Study: PhD, biological sciences, University of California, Santa Barbara; Postdoctoral Studies: Mayo Clinic, Rochester, Minnesota
Postdoctoral Studies: Mayo Clinic, Rochester, Minnesota
Connect with Penn State Bone and Joint Institute on Doximity

