Chemoembolization and Radioembolization for Difficult Liver Tumors; Choice May Depend on Blood Flow
Two-thirds of primary liver cancer patients and 90 percent of secondary liver cancer patients have inoperable tumors that are unresponsive to systemic chemotherapy and may benefit from transarterial chemoembolization or radioembolization with yttrium-90 (Y-90), both minimally-invasive, palliative procedures. Although research shows transarterial chemoembolization and Y-90 are usually equivalent treatments for most intermediate-stage hepatocellular carcinomas (HCCs)1, Heart and Vascular Institute considers tumor blood supply when deciding which therapy would be most beneficial for a specific patient.
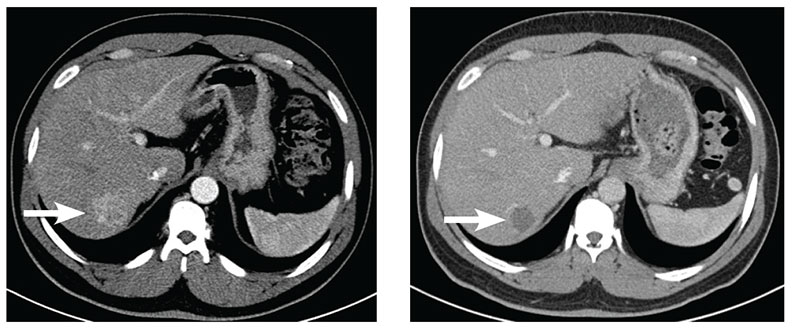
Image on the left: Pre-chemoembolization axial, contrast-enhanced CT scan at the level of the liver. White arrow shows a hypervascular lesion (33.8 mm x 31.6 mm) in segment 7 of the liver consistent with hepatocellular carcinoma.
Image on the right: Post-chemoembolization axial, contrast-enhanced CT scan at the same level as the previous image. White arrows show the treated lesion (22.4 mm x 24.0 mm) in segment 7. The lesion is no longer enhancing and has decreased in size.
Chemoembolization involves injection of 100-300 micron microspheres loaded with chemotherapy to block arteries that feed tumors, and to deliver chemotherapy directly to the tumor, sparing normal liver tissue. The team at Penn State Health Milton S. Hershey Medical Center tends to use chemoembolization for HCC and other hypervascular tumors that draw more blood flow than the surrounding tissue, including neuroendocrine tumors. “Any embolic material put into the arteries supplying the tumor and adjacent parenchyma is more likely to distribute in the area of higher blood flow,” explains Allene Salcedo Burdette, MD, assistant professor of radiology, surgery and medicine.
Radioembolization delivers a targeted, high dose of radiation, 20-60 micron microspheres, via smaller delivery. The microspheres act as Y-90 delivery devices and enable radiation to distribute more distally “and more widely in hypovascular tumors that don’t attract a great deal of blood flow, compared to a tumor that draws a lot of blood,” Dr. Burdette adds. Mapping angiography with a radiotracer agent, performed prior to radioembolization, determines if Y-90 is safe for individual patients by mimicking Y-90 distribution. If the tracer is found in surrounding, non-hepatic tissues or an excessive amount enters the lungs, Y-90 therapy is deemed unsafe. At Hershey Medical Center, Y-90 is typically used for radioembolization of hypovascular metastases in the liver, such as colorectal cancer.
Candidates for both treatments include those with primary liver disease and those with metastatic liver disease, who may have diminished liver function from multiple rounds of chemotherapy or multiple lesions that begin to replace the normal hepatic parenchyma. “A risk of embolization of the non-tumorous liver tissue is liver failure,” Dr. Burdette notes. “The more abnormal the liver function, the higher the risk of liver failure.”
Side effects of chemoembolization include flu-like symptoms and pain lasting from a few days to a few weeks; however, some patients have no adverse effects. Y-90 patients tend to better tolerate treatment, yet experience significant fatigue.
In a study of 291 Y-90 patients, survival differed significantly by cancer stage and liver function, ranging from 7.7 to 26.9 months.2 A multi-institutional trial of chemoembolization in 112 patients with inoperable HCC—whose mean survival is three to six months—showed one-year and two-year survival rates at 82 percent and 63 percent, respectively.3
“Chemoembolization and Y-90 are minimally invasive options, and have real effect in prolonging patients’ lives,” Dr. Burdette emphasizes.

Allene S. Burdette, MD
Assistant Professor, Radiology, Surgery and Medicine
Interventional Radiologist
Email: asb7@psu.edu
Fellowship: Vascular and interventional radiology, University of Utah Medical Center, Salt Lake City, Utah
Residency: Diagnostic radiology, Penn State Health Milton S. Hershey Medical Center, Hershey, Pa.
Medical School: University of Kentucky College of Medicine, Lexington, Ky.
Connect with Allene S. Burdette, MD, on Doximity
References:
- Fidelman N, Kerlan RK Jr. Transarterial Chemoembolization and (90) Y Radioembolization for hepatocellular carcinoma: review of current applications beyond intermediate-stage disease. AJR Am J Roentgenol. 2015 Oct;205(4):742-52.
- http://www.medscape.com/viewarticle/719102#vp_4. Accessed March 1, 2016.
- Douglas E. Ramsey, AB; Jean-Francois H. Geschwind, MD. Chemoembolization of unresectable hepatocellular carcinoma: a review. Appl Radiol. 2004;33(3).
